After entering the spacious VanVeller Lab, it is immediately evident that this dynamic workspace is where chemists collaborate, discover and conduct award-winning research.
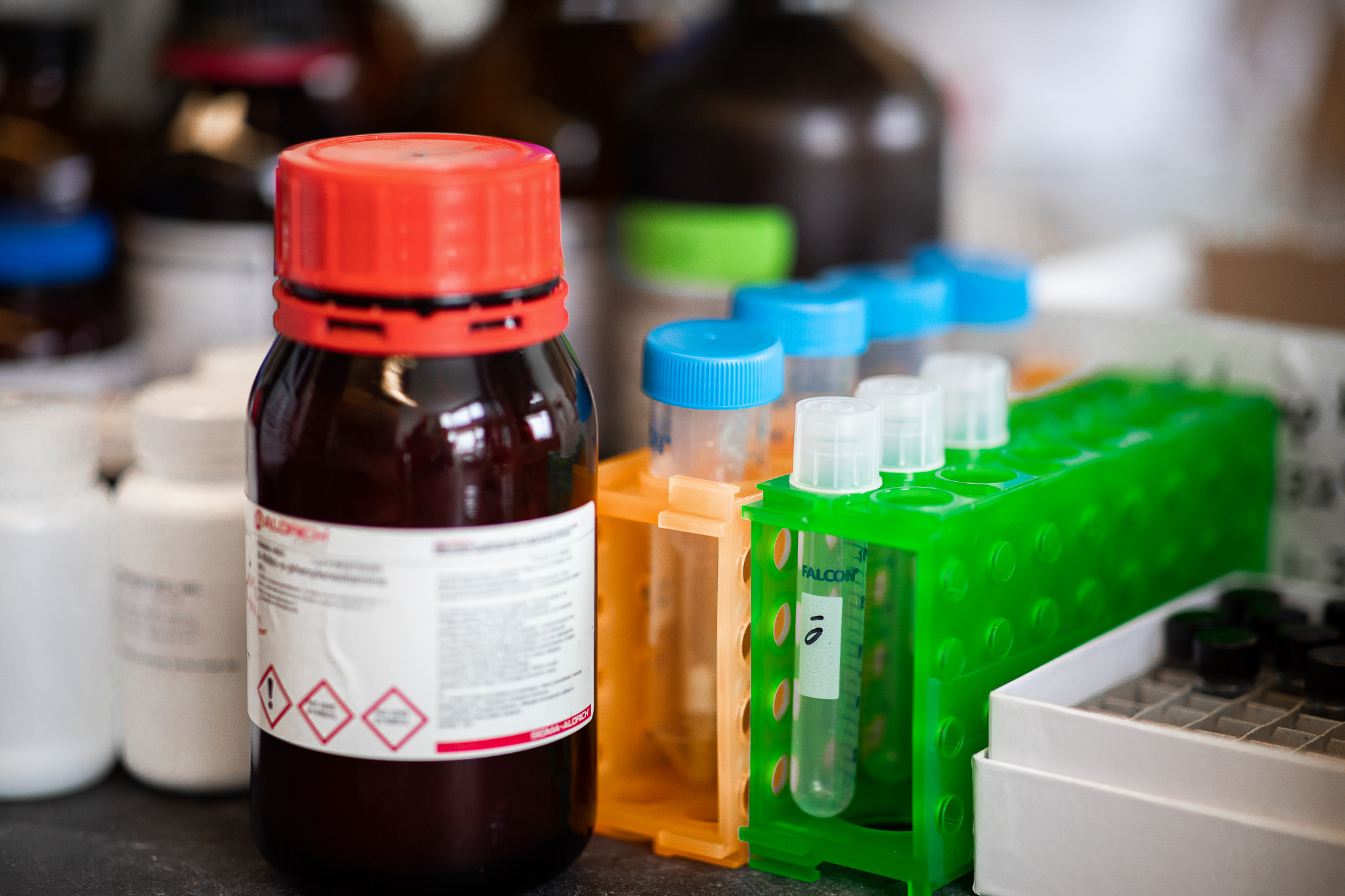
From the smallest of hollow pipettes to the heaviest of metal tanks filled with liquid nitrogen—the traditional trappings of a chemistry research lab fill the shelves and crowd the countertops.
Workstations teem with glass beakers, flasks and rows of test tubes that contain red, orange and green liquids. A trio of jars filled with glowing, iridescent powders signals that the inventive work of photochemistry research occurs here.

Brett VanVeller, assistant professor of chemistry, is the principal investigator who leads a diligent team of six student researchers—five graduate students and one undergraduate. VanVeller joined the Department of Chemistry in 2014.
"Brett was outgoing and obviously enthusiastic about science from the day he first interviewed on campus,” said William Jenks, professor and chair of the chemistry department. “It shows when he talks about his work and it shows when he’s teaching. VanVeller is ‘all in’ when it comes to making sure that we do right by Iowa State students.”
There is an air of collaboration and creativity among the lab members who dart from workstation to workstation in crisp, white lab coats—jotting down notes, recording new discoveries and sharing new findings.
“The students who work in my lab will be the first to tell you that the most important things they learn happen outside of the classroom.”
“Chemistry is amazing because it explains how and why everything works. If you understand how atoms and molecules work, it turns out that everything is made of atoms and molecules,” VanVeller said. “Every day, the excitement of this central science plays out in the lab. There’s so much to learn and experience here.”
Being trapped behind a desk or memorizing reams of information never appealed to VanVeller. He prefers actions and experiences. Investigating, researching and developing new luminescent molecules are what gets him out the door every morning.
“I also try to impart an excitement for experiential learning to all of my students—whether they’re enrolled in my chemistry classes or working in the lab," said VanVeller.
Illuminating research
VanVeller’s research focus is photochemistry, a cutting-edge branch of chemistry that explores the chemical effects of light.
The potential and promise of his photochemistry research recently earned VanVeller a 2019 Faculty Early Career Development (CAREER) award. This honor is the National Science Foundation’s (NSF) most prestigious recognition in support of early-career faculty members who serve as exceptional teacher-scholars through outstanding research, teaching excellence and the integration of education and research.

“Investment in exemplary faculty members early in their careers is key to achieving the National Science Foundation's mission to enhance the United States' science and engineering ecosystem," said NSF Director France Córdova. “We look forward to these discoveries and innovations that will advance the frontiers of scientific understanding."
Caroline Warner (Ph.D. ’21 chemistry) says that working as a student researcher in an award-winning photochemistry lab has been “an education in itself.” This former high-school chemistry teacher notes that her most memorable research project involved fireflies.
Yes, fireflies.
Under VanVeller’s leadership, Warner and other team members worked to produce alternative forms of luciferin, the molecule that gives fireflies their glow. When your eye catches a dark field dotted with blinking fireflies, you’re witnessing the handiwork of luciferin.
Instead of allowing the synthetic luciferin to emit its standard, yellow-green light, Warner attempted to chemically persuade the luciferin to produce different colors of light.

“It’s pretty amazing to see how organic chemistry dictates the firefly’s glow and also how we can manipulate that chemistry in the lab to change the color of the light,” Warner said.
So why is it necessary to alter a firefly’s natural shimmer?
“The biochemical machinery responsible for the firefly’s glow is an important biotechnology that is used to study numerous models of disease,” VanVeller said.
While working in the VanVeller Lab, Warner learned that red light is healthier for human cells; and it can also be detected at much deeper depths through skin and tissue.
“For example, when you shine a red flashlight through your hand, you can see red light through your hand. But you aren’t able to see blue light,” she said.
"It’s pretty amazing to see how organic chemistry dictates the firefly’s glow and also how we can manipulate that chemistry in the lab to change the color of the light.”
VanVeller notes that although many informative insights were gleaned from the luciferin research, the lab has transitioned to other projects that are driving his research in new and compelling directions.
“We learned a lot about what works and what doesn’t work with the luciferin project," he said. "Now we’re parlaying that knowledge into many exciting areas, one of which is fluorescence,” he said.
Tagging proteins with light
What happens when you infuse fluorescence—or molecules that glow—into targeted proteins within the human body? Those proteins light up, of course.

“When your teeth glow under a black light in a bowling alley, for example, your teeth are absorbing the energy from the black light and then emitting that light at a slightly different color. That’s fluorescence,” VanVeller said. “Proteins can be tagged with the same kind of fluorescent molecules, making what used to be undetectable, now detectable.”
When proteins glow with fluorescence, medical professionals are better able to view, track and analyze them. This is especially helpful when proteins grow into harmful plaques, or amyloids. The spread of these problematic amyloids is the root cause of degenerative conditions, such as Parkinson’s and Alzheimer’s diseases.
“When fluorescence is used to light up amyloids in the brain, doctors can detect disease earlier and begin treatment during those critical first stages of disease,” VanVeller said. “Rapid screening tests and medical diagnostics can revolutionize patient health—and photochemical research is key to those advances.”
“Every day, the excitement of this central science plays out in the lab. There’s so much to learn and experience here.”
VanVeller added that the potential contributions of photochemistry are “endless” and “could lead to breakthroughs in chemical synthesis, materials fabrication and the targeted delivery of drugs.”
“This is the beauty of photochemistry research and lab work in general,” he said. “We’re constantly experimenting, evolving and asking questions, which is the best way to learn, discover and innovate.”
VanVeller’s affinity for experiential learning is evident today and dates back to decades before he stepped foot inside a chemistry lab.
Catalysts for change
Throughout his grade school years, VanVeller’s parents observed their son's intense curiosity about the natural world. His interest in space, astronomy and zoology stood out, as did his penchant for science-focused books that he routinely checked out from the local library.
He was always asking questions.

“My mother was a very smart woman who always encouraged me to learn and ask questions. She would have been an amazing scientist,” VanVeller said. “My father was constantly building and fixing things, and he never stopped pulling me into his ongoing projects. My parents always encouraged me to follow my passions.”
While science books provided quiet learning opportunities, VanVeller’s parents felt compelled to offer their son hands-on learning experiences that brought science to life. They planned a daytrip to the nearest science center—which was three hours away.
“The experience was really impactful; it ignited my fascination with how the complexity of matter and life emerges from the relative simplicity of atoms and molecules,” VanVeller said. “I’ll always remember that trip as my first step on the path to becoming a scientist.”
Inspiring tomorrow’s chemists

VanVeller’s own history has taught him that today’s awestruck young learner could become tomorrow’s creative chemist. In response, VanVeller has spearheaded a variety of science activities designed for young children. He often uses the summer term to educate kids about science and introduce them to chemistry.
In addition to visiting his daughters’ classrooms with liquid nitrogen demonstrations and visual exercises that teach the fundamentals of chemical bonds, VanVeller also hosted 24 campers from the Science Center of Iowa’s SciGirls program.
VanVeller transformed these girls into amateur scientists for a day. They made super slime and noshed on ice cream made with liquid nitrogen. Each camper took home a glass bottle that they plated with silver, a memorable souvenir from which they learned about the states of matter.
“It’s important to get out into the world and show people the excitement of science through fun demonstrations and memorable experiences,” VanVeller said. “The students who work in my lab will be the first to tell you that the most important things they learn happen outside of the classroom.”
From first graders to graduate students, VanVeller’s teaching skills have impacted many. His talents as an educator have garnered national honors, including a 2018 Cottrell Scholar Award. This prestigious award recognizes outstanding educators who are also excellent researchers and academic leaders in their field.
In 2017, he earned a Cassling Family Faculty Award for Early Achievement in Teaching, as well as an Early Achievement Teaching Award from the College of Liberal Arts and Sciences at Iowa State University.
Unbreakable bonds

Great chemistry in the VanVeller Lab isn’t only found in the lab notes and research results. It’s also infused within the connections and relationships between the lab members who work on this close-knit research team.
VanVeller’s charismatic and communicative leadership style fosters a creative, team-oriented atmosphere in the lab.
Bryan Lampkin (Ph.D. ’20 chemistry) arrived at Iowa State in 2015 and was drawn to VanVeller’s photochemistry research. He hoped to work in his lab, but he lacked the chemistry and biology knowledge that was needed to perform the research.
“I would not be where I am today without Dr. VanVeller's influences and the incredible experiences I had working in his lab. I learned so much from him.”
“Dr. VanVeller said five words that changed my career and my perspective on scientific research,” Lampkin said. “He told me, ‘Just pick up a textbook.’”
“Those words inspired me to take control of my learning. Now, I don’t complicate things,” Lampkin said. “I identify what needs to be done and I figure out how to do it. It can be as easy as picking up a book. It’s all very empowering.”
After heeding VanVeller’s advice, Lampkin worked hard to master a variety of subjects. His due diligence earned him a spot in the VanVeller Lab, where he has worked since 2015. Lampkin is also a teaching assistant for VanVeller’s organic chemistry class and an occasional guest lecturer for his second-year organic chemistry class.

“I’m preparing for a career as a professor,” Lampkin said. “There is a massive learning curve when you begin work in a new lab. I overcame it with Dr. VanVeller’s help. He coaches to the level of the student, and I hope that I’m able to inspire my own students as Dr. VanVeller has inspired so many of us.”
Warner agrees that the lessons learned from working with VanVeller extend far beyond covalent bonds and formulas.
“I was struggling with physical organic chemistry. Dr VanVeller organized a ‘chemistry boot camp’ during his free time in the summer and it really helped,” Warner said. “Not only did I learn about organic chemistry, I learned the importance of being a generous and patient teacher.”

VanVeller provides his students with a solid foundation of chemistry knowledge as well as opportunities to hone their lab skills. These valuable experiences have helped students to land scholarships, fellowships and even their dream jobs.
Brittney Ross (’16 chemistry) is a researcher who is working on her Ph.D. at the University of Chicago’s Molecular Engineering Program. Her research, which is conducted in the Esser Kahn Lab, could someday lead to single-dose vaccines and eliminate the need for booster shots.
"Dr. VanVeller is an incredibly inspiring, charismatic and enthusiastic young scientist,” she said. “I would not be where I am today without Dr. VanVeller's influences and the incredible experiences I had working in his lab. I learned so much from him.”
The future of the VanVeller Lab

The VanVeller Lab will remain focused on photochemistry research. Undoubtedly, his team will continue to explore many offshoots of this compelling branch of chemistry.
Currently, the team is producing new classes of peptides and biomolecules.
“This really is the future thrust of the lab,” VanVeller said. “We’ve had early success with peptides and molecules that can be manipulated with light. We’re able to modulate their response to light and it’s very exciting and promising work.”
VanVeller points out that Lampkin, who originally lacked a biochemistry background, is currently spearheading some of the grant writing and research projects involving peptide modeling in the lab.
“Bryan has surpassed me when it comes to peptide biology, database searching and bioinformatics,” VanVeller said. “The student has become the expert, and Bryan’s knowledge is fueling new discoveries in our lab. Bryan really ran with it. It’s awesome when your students become experts in their own right.”
VanVeller is thrilled that members of the VanVeller Lab are learning, growing and using their experiences to build out their dreams and advance their careers.

“That’s really the goal of teaching students and working with them in a lab, isn’t it?” VanVeller said. “You provide the experiences, and students then use those opportunities to cultivate their own success stories. That’s education, and it’s the best thing about my job."
